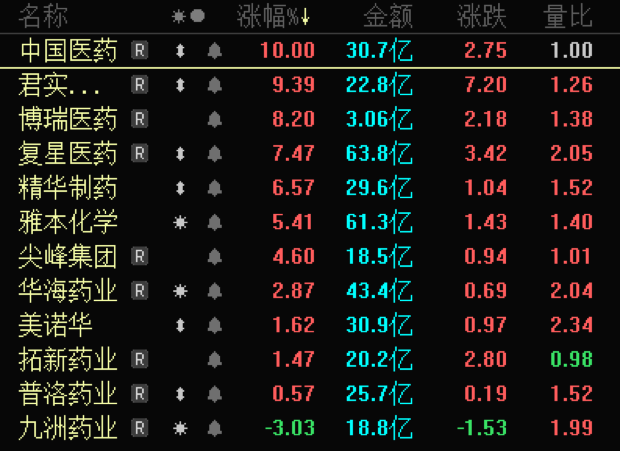
Today (17th) the new crown medicine sector is strong,Fosun Pharma、Essence Pharma、Pinnacle Group、Huahai Pharmaceutical、Minovar、Pro Pharma, Kyushu Pharmaceutical once hit the daily limit. As of the close,Chinese medicinedaily limit,Junshi Biology、Borui Medicine、Fosun Pharma、Essence Pharma、Masamoto Chemistryrose more than 5%.
The direct catalyst is that it was reported on the 17th that the Geneva Medicines Patent Pool (MPP) organization announced that it had signed agreements with 35 pharmaceutical companies to allow their productionPfizerNirmatrelvir, one of the components of the new crown oral drug Paxlovid, is a raw material drug or preparation, including Shanghai Desano,Huahai Pharmaceutical、Pro Pharma、Fosun Pharma、Jiuzhou Pharmaceuticalthese five Chinese companies.
Paxlovid asPfizerThe original research is the new crown drug with good curative effect in the world.Today’s daily limitChinese medicineIs Paxlovid’s agent in mainland China.
As of press time, MPP’s official website has not announced the above news, and no relevant pharmaceutical companies have confirmed it.
but,PfizerThe signal for an open license has been sent before.On January 18, local time, Pfizer announced that it had signed a voluntary licensing agreement with MPP to help expand its accessibility in 95 low- and middle-income countries, which account for about 53% of the world’s population. But the news did not reveal which companies were involved.
The diagnosis and treatment plan is now important to adjust the active layout of domestic manufacturers
At present, the Omicron strain has replaced the delta strain as the main epidemic strain, and the patients with the Omicron strain are mainly asymptomatic infected persons and mild cases, and the new crown oral drug is an important treatment method. In view of this, the idea of the prevention and control of the new crown in the future is changing.
On the evening of the 15th, the National Health and Medical Commission issued the “New Coronavirus Pneumonia Diagnosis and Treatment Plan (Trial Version 9)”.Pfizer’s oral drug Paxlovid and Tengsheng Biopharma’s ambavirumab/romisevirumab were written into the recommended treatment regimen. In addition, the two sessions also proposed to accelerate the research and development of new crown drugs.
At the same time, the progress of domestic pharmaceutical companies in oral medicine is expected. Since March, many companies have spoken out and disclosed the staged results:
Junshi BiologyOn the 16th, it was announced that the Phase I data of the new crown oral drug VV116 jointly developed with Wangshan Wangshui was “satisfactory”, and the Phase III clinical study has been launched;Pioneering PharmaceuticalsThe relevant person in charge said in an interview with the media recently that three global multi-center Phase III clinical trials of proclutamide for the treatment of new crowns are currently in active progress, and will be announced in late March 2022 no later than early April. line data.
MinovarOn the 15th, it disclosed the relevant matters for the filing and approval of the “new project of the anti-new crown drug naimatevir API and its key intermediates with an annual output of 60 tons”. It plans to add 90 tons of SM1 production capacity and 120 tons of SM2 production capacity. The company has now delivered 5 tons of SM1 and 10 tons of SM2, and there are dozens of tons of orders in negotiation.The reporter of “Science and Technology Innovation Board Daily” called todayMinovarsecuritiesMinistry, a company official said, “The customer unit cannot be disclosed, but the company is indeed in the MPP supply chain.” It also revealed that the order negotiation is progressing smoothly.
Three investment directions are in sight
Specific to investment opportunities, comprehensiveanalystThere are three main points of view:Enterprises authorized by overseas generic drug MPP, domestic R&D and innovation enterprises with advanced progress, and potential upstream suppliers of the new crown drug industry chain.
DongguansecuritiesAnalyst Wei Hongmei said that the domestic new crown small molecule drugs are expected to be launched in the future, and the overall market prospect is relatively broad.It is recommended to pay attention to the rapid development of new crown small molecule drugsJunshi Biology;CDMO companies deeply involved in the global COVID-19 small molecule supply chainWuXi AppTec、Aleigh、Porton sharesWait.
people’s livelihoodsecuritiesAnalyst Zhou Chaoze expects that MPPannouncementPaxlovid generic drug manufacturers will be launched in the near future. The huge global market will bring supply opportunities for Chinese API companies in the industry chain. At present, the price of key upstream intermediates is still at a high level. Companies with relevant technology and capacity reserves are expected to bring Good profit elasticity, recommended attentionHuasoft Technology、Huahai Pharmaceutical、Jiuzhou Pharmaceutical、Shinwasei、Jincheng Medicine、Lepu Medical、Pro PharmaWait.
Zheshang SecuritiesAnalyst Sun Jian releasedResearch reportIt is said that overseas new crown specific drug intermediates and APIs are expected to become global large-tonnage varieties. Based on strong synthesis technology, relatively complete chemical facilities and advanced production capacity advantages, some companies are expected to continue to benefit from Chinese APIs and CDMO companies. The supply chain of new crown special drugs under the global industrial division of labor. In terms of CDMO, it is recommendedWuXi AppTec、Aleigh、Porton shares、Jiuzhou PharmaceuticalPuluo Pharmaceutical and other companies; in terms of APIs, it is recommended to pay attention toTianyu sharesHuahai Pharmaceutical,Orient、Aoxiang Pharmaceutical、Borui Medicineand other companies.
(Article source: Financial Associated Press)