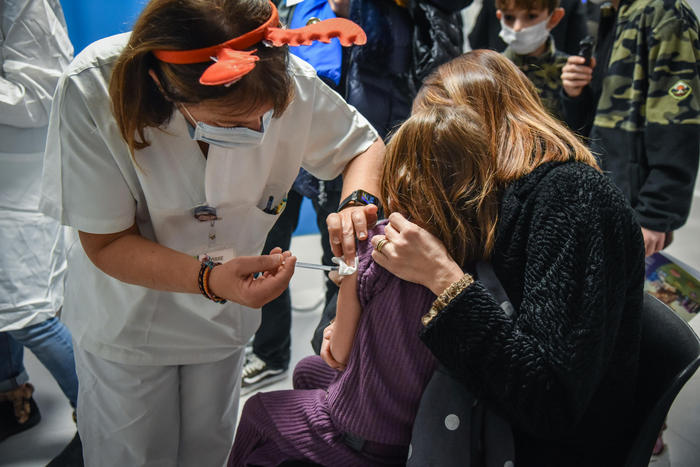
One day after the opening of the administrations, 15 thousand children between 5 and 11 years old took the first dose of vaccine against Covid. The data is published on the government website and is updated this morning. Out of a total audience of 3,656,069 15,063 children were vaccinated. Instead, 104,135 are those who have contracted Covid and are healed.
In children who will turn 12 years of age after having received a first dose of Comirnaty vaccine (BioNTech / Pfizer) in the formulation of 10 mcg / dose in 0.2 mL, it is possible to complete the primary course of vaccination, in compliance with the scheduled times. , administering a second dose of vaccine with the same formulation as the first dose. This was specified by the circular of the Ministry of Health ‘Clarifications regarding the indications for the use of the Comirnaty vaccine (BioNTech / Pfizer) for the age group 5-11 years’. The second dose is scheduled three weeks after the first.
Furthermore, it is emphasized in the circular signed by the Director of Prevention of the Ministry of Health Gianni Rezza, “in case of previous confirmed SARS-CoV-2 infection or in case of infection following the administration of the first dose, the indications provided in this regard remain valid. for individuals aged 12 years or older “.
Also for vaccinated with ReiThera booster at 5 months
Also for subjects vaccinated in Italy in the context of the anti-Sars-CoV2 vaccination clinical trial called COVITAR, with the ReiThera vaccine, it will be possible to proceed with the administration of a booster dose of m-RNA vaccine, in dosages authorized for the same, at least 5 months after the last dose received.
The administration of this booster dose is suitable for the issue of the related Covid-19 green certification. The new circular of the Ministry of Health provides for this ‘Indications for vaccinated in clinical trials with experimental anti-SARSCoV-2 / COVID-19 vaccines in Italy’.
Booster from 28 days to 6 months in vaccinated Takis
In relation to the subjects participating in the Covid-eVax-Takis trial and who at the opening of the codes did not result in the placebo arm, “the provisions indicated for subjects vaccinated abroad with a vaccine not authorized by EMA apply”. The new circular of the Ministry of Health provides for this. Subjects who received the Takis experimental vaccine can therefore receive a booster dose with mRNA vaccine, starting at 28 days and up to 6 months after completion of the primary course. After 6 months, as well as in case of failure to complete the cycle, a primary vaccination cycle with mRNA vaccine can be proceeded.
.