- Pallab Ghosh
- BBC Science Correspondent
image source,TEK IMAGE/SCIENCE PHOTO LIBRARY
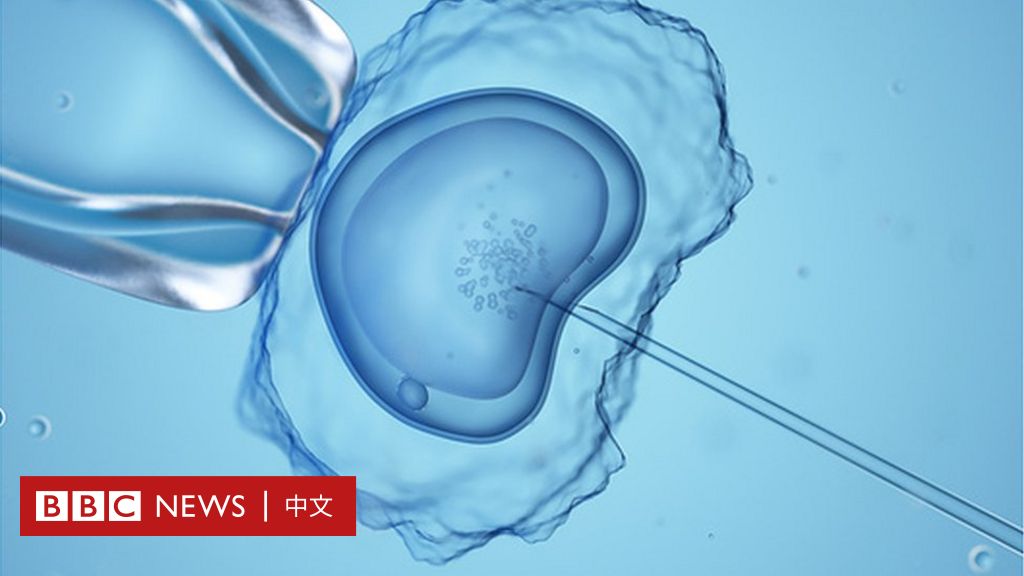
Gene-editing technology could potentially be used to treat many genetic conditions
A British expert has warned that China’s new rules for human gene editing are not perfect.
Dr Joy Zhang, a science ethics expert at the University of Kent in the UK, said the authorities were involved in “improper regulations”.
The rules were updated five years ago after a Chinese researcher said he had produced the first genetically altered baby.
China says the new law complies with international rules.
They are aware of the requirements for ethical accreditation, monitoring and verification, but experts worry that these regulations may not apply to private industry.
Dr Zhang, who was one of the key speakers at an international conference on human gene editing in London, told the BBC her biggest concern was that the new measures did not address long-standing and growing problems in the private sector, those outside the general scientific community. activity.
She said the new rules could be overwhelmed amid China’s burgeoning innovation scene.
Gene editing is a new technology that allows researchers to make precise changes to DNA. Researchers believe gene editing could be used to correct many genetic diseases.
But the technique is controversial because it could permanently alter a person’s genetic makeup and pass the genetic changes on to future generations.
controversial trial
Dr He Jiankui of Shenzhen, China, said five years ago that he had created the world‘s first genetically altered babies – twin girls. The news shocked leading scientists in the field of genetic research in various countries.
Dr He said the baby girls’ DNA was edited early in the embryonic stage so that they would be resistant to HIV.
In 2019 Dr He was fined and sentenced to three years in prison. Except for He Jiankui, no one else had contact with the twins. At a recent scientific meeting, Dr. He said they were in good health.
News broke last month that Dr. He plans to set up a clinic in Hong Kong after his release from prison to use gene-editing technology to find a cure for a rare genetic muscle disorder in children known as Duchenne Muscular Dystrophy.
Hong Kong immigration officials said they had revoked Dr He’s entry permit after discovering that he had a criminal record.
The new rules close loopholes that would allow He Jiankui to evade the rules, which he has previously used to apply for human trials in hospitals, such as drug trials. The updated regulations cover all research institutions and all aspects of the human body, including human tissues, organs and embryonic cells.
image source,He JiankuiLab
In 2019, Dr. He Jiankui was fined and sentenced to three years in prison after claiming to have created gene-edited babies.
The organizer of the International Conference on Human Gene Editing in London is Prof Robin Lovell-Badge of the Francis Crick Institute in the UK, where the conference was held. Professor Robin Lovell-Bage said that there are too many secrets in China studies that attract his attention.
He said he fully understands why China wants to take the lead in this technology, but some areas of research require special attention, gene editing being one such field.
“It needs to be properly managed and supervised, and I’m afraid that hasn’t been done there yet.”
Speaking at the summit, Dr. Yangin Peng of the Chinese Academy of Sciences said the Chinese government had “accelerated” the development of laws and regulations on gene editing.
“China has significantly tightened legislation and regulations, permanent, genetic genetic alterations are banned, management has taken a more cautious approach, and our laws are in line with international rules,” he said.
image source,SPL
Dr. Françoise Baylis, a bioethicist at Canada’s Dalhousie University, would like more details on China’s updated rules.
“I see (in the updated regulations) that research should be done in accordance with ethical principles. I want to know according to which ethical principles, where those principles are formulated, and whether they will be challenged,” she said.
She also said that regulating the private sector is not a problem unique to China. “We have the same problem in North America, so I think it is wrong to only target China.”
Dr. Piers Millett is from the International Biosafety Program in Washington. He acknowledged that “China may be ahead of the game in terms of rule revisions in this area.”
image source,SEBASTIAN KAULITZKI/SCIENCE PHOTO LIBRARY
Gene editing can precisely change genes at the embryonic stage of cells
Many researchers have wondered how Dr. He Jiankui would be allowed to return to clinical research after serving his sentence.
Dr. Joey Zhang said, “Like many others, I have been wondering whether someone or an institution in China is supporting or protecting He Jiankui.”
But she said now she thinks “we’re dealing with an example of poor regulation”.
This leads her to believe that “without clarifying the He Jiankui case, recent claims of good management are hypocritical.”
“I’m not so worried about what He Jiankui is going to do, I’m more worried about what the Chinese authorities are doing.”