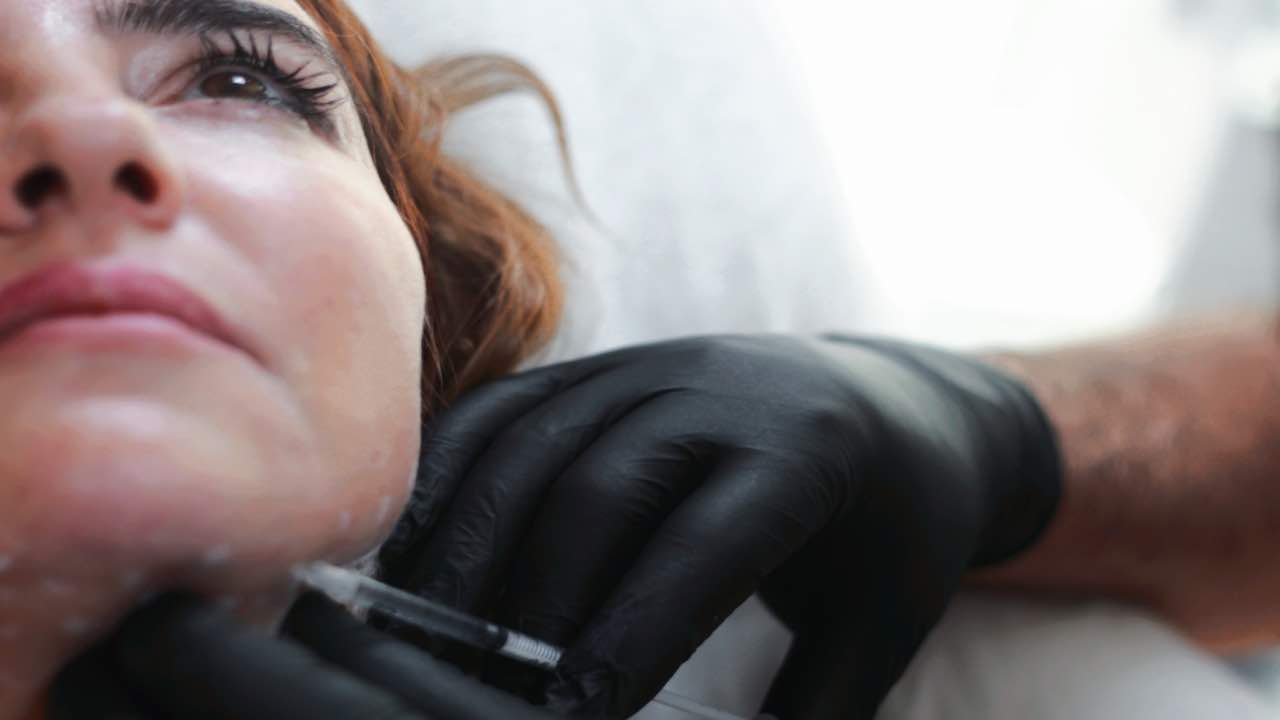
The best known use of hyaluronic acid is the aesthetic one, with small infiltrations that have the purpose of attenuating the signs of aging, but this molecule is widely used in the medical field also for other treatments. However, too many side effects were noted and the product had to be recalled.
A recent safety notice released by the pharmaceutical company Compensationwhich produces hyaluronic acid for therapeutic purposes, has in fact clarified what are the problems that a patient who has been given doses of this molecule can face and clarifying that he has therefore taken steps to withdraw the product from the market.
Hyaluronic acid for medical purposes, watch out for side effects
Lo Hyalubrix a solution based on hyaluronic acid produced by the pharmaceutical company Fidia and sold in pre-filled disposable syringes for medical use. It is usually used for patients affected by joint problems – such as degenerative arthropathy of the knee, shoulder, hip and so on.
This is a solution that is usually reached when the problem becomes chronic (for which the common analgesics stop having effect) or in the case in which there is a certain threshold of intolerance to other types of anti-inflammatory medicines.
After several monitoring of the use of this device, however, several side effects were found and the drug had to be withdrawn from the market. In particular, the subjects who used the Hyalubrix experienced pain and swelling at the level of the joint for which they started the treatment.
Fortunately, these symptoms are not particularly serious but widespread enough to require precautionary measures to preserve the health of patients – as also reported in the press release issued and mentioned above.
Hyaluronic acid for medical purposes, what is it
As mentioned above, many know the benefits of hyaluronic acid in the aesthetic field, but this type of molecule is also widely used for medical purposes and – as explained – especially in the treatment of pain in the event that the patient cannot take analgesics. (because it has become resistant or intolerant to the active ingredient).
Obviously, these are two completely different types of treatment. In the case of Hyalubrix we are dealing with an obviously sterile viscoelastic solution produced with hyaluronic acid sodium salt derived from bacterial fermentation. It is also a high molecular weight concentration – which means, in a nutshell, that the size of the molecules does not allow it to pass beyond the epidermis.
As anticipated, the device, before being withdrawn from the market, was sold in a pre-filled syringe containing 30mg / 2 ml of solution, sterilized in a humid environment, and sealed in a blister in order to make the device disposable.