The most diverse therapies for treating brain diseases have a common problem: they have difficulty achieving their goal. The blood vessels that run through the brain have a special lining that is so densely packed with cells that only very small molecules can pass through. This blood-brain barrier “acts like a seal” that protects the brain from toxins or other harmful substances, says molecular biologist Anne Eichmann of Yale University. But the seal also keeps most medications out. For decades, researchers have been working on methods to get drugs through the blood-brain barrier.
Advertisement
Neuroscientists at West Virginia University have now succeeded in using focused ultrasound to open the blood-brain barrier and improve the delivery of a new Alzheimer’s drug. As they reported in the New England Journal of Medicine in mid-January, this accelerated the breakdown of the sticky plaques that are linked to some of the cognitive and memory problems in Alzheimer’s patients by 32 percent.
In the West Virginia study, three people with mild Alzheimer’s disease received monthly doses of aducanumab, a lab-made antibody administered via infusion. First approved in 2021, this drug helps eliminate beta-amyloid, a protein fragment that clumps together in the brains of Alzheimer’s patients. The drug’s approval was controversial, and it is still not clear whether it actually slows the progression of the disease.
After the infusion, the researchers treated certain regions of the patients’ brains with focused ultrasound, but only on one side. This allowed them to use the other half of the brain as a control. PET (positron emission tomography) scans showed a greater reduction in amyloid plaques in the ultrasound-treated regions than in the same regions on the untreated side of the brain. This indicates that more antibodies reached the brain on the treated side.
Aducanumab clears the plaques even without ultrasound, but it takes a long time. Perhaps also because the antibody has difficulty penetrating the brain. “Instead of administering the therapy intravenously for 18 to 24 months to achieve the reduction in plaques, we want to see if we can achieve this reduction in a few months,” says Ali Rezai, a neurosurgeon at the Rockefeller Neuroscience Institute at West Virginia University and Author of the new study. Reducing the time it takes to clear these plaques could help slow the memory loss and cognitive problems that characterize the disease.
Bubbles against barrier
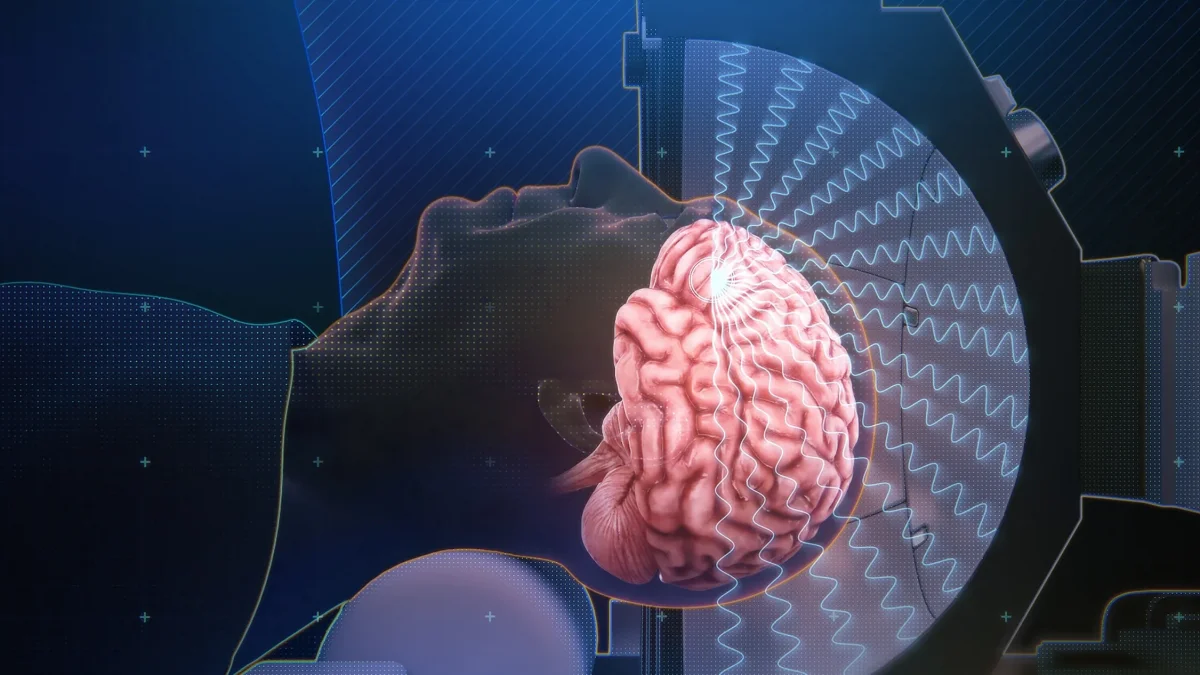
The device for aligning and emitting the ultrasound waves consists of a magnetic resonance tomograph (MRI) and a helmet equipped with ultrasound transducers. It was developed by a company called Insightec. It is FDA-approved, but for a very different purpose: to help stop tremors in Parkinson’s patients by creating lesions in the brain. To open the blood-brain barrier, “we inject patients with microbubbles intravenously,” says Rezai. These tiny gas bubbles, commonly used as contrast agents, travel through the bloodstream.
Using MRI, researchers can direct the ultrasound waves to very specific parts of the brain, “with millimeter precision,” says Rezai. When the waves hit the microbubbles, they begin to expand and contract, physically pushing apart the tightly packed cells that line the brain’s capillaries. “This temporary opening can last up to 48 hours, meaning that during these 48 hours therapeutics can better penetrate the brain,” he says.
Focused ultrasound has been researched for years as a method for opening the blood-brain barrier. However, this is the first time it has been combined with Alzheimer’s therapy and tested in humans.
To home page